SaphenionScience – 90 months vein glue in 1365 cases
SaphenionScience – 90 months vein glue: Treatment with Cyanoacrylate glue is using for a long time. Since 1949 the chemical compound is known, first being used in operative medicine in the early 60s as a Tissue adhesive or replacement of wound sutures.
SaphenionScience – 90 months vein glue: Nearly all operative disciplines are using cyanoacrylate i.e dermatology, ophthalmology, orthopedics, surgery, orthodontics, interventional radiologists, vascular surgery. The risk of allergic reaction is not real – we have found two reports in pubmed about an allergic reaction after therapy with cyanoacrylate vein glue. This is all the more astonishing since more than 100,000 patients have now been treated with vein glue worldwide! We have not seen ourself an allergic reaction all over the time of 90 months.
In the guidelines of the German Society of Phlebology (vein medicine), the vein glue has now been included in the treatment recommendations, in the United States, the VenaSeal has been declared a compulsory insurance.
The CE approval for sealing veins was in 9/2011 for Europe, the FDA approval in the US followed in 2/2015. The FDA states in its documentation: VenaSeal® is bio-compatibel and bioresorptive, non-carcinogenic and non-allergenic!
SaphenionScience – 90 months vein glue in 1365 cases
Until February 2th. 2020 we sealed GSV in 1750 cases, SSV in 655 cases, VSAL in 95 cases, VSAM in 71 cases, Femoropopliteal vein in 23 cases, Giacomini`s vein in 2 cases.
Treatment of one truncal vein in 393 cases, 760 patients received treatment for two truncal veins simultaneously, in 170 cases we treated three truncal veins. In 36 cases patients have got treatment of four truncal veins, in 5 case 5 truncal veins. One patient was treated for 6 truncal veins and an aneurysma in a single session (summary: 1365 patients).
In 16 cases we treated truncal veins with ulcera crures: they healed 2 to 12 weeks after. We also treated patients with HIV, hepatitis, multiallergenic constitution or Parkinson without any pathological reaction or side effects.
In one case we treated a patient who was known to be allergic to a substance used in cyanoacrylate production. After asking the manufacturer of VenaSeal®, the therapy was carried out without any problems.
Nearly all procedures carried out under sedation (dormicum i.v.), 175 patients without anesthesia (12,8%), one patient performed pain acupuncture in itself.
SaphenionScience – 90 months vein glue on 2595 truncal varicose veins: Our results
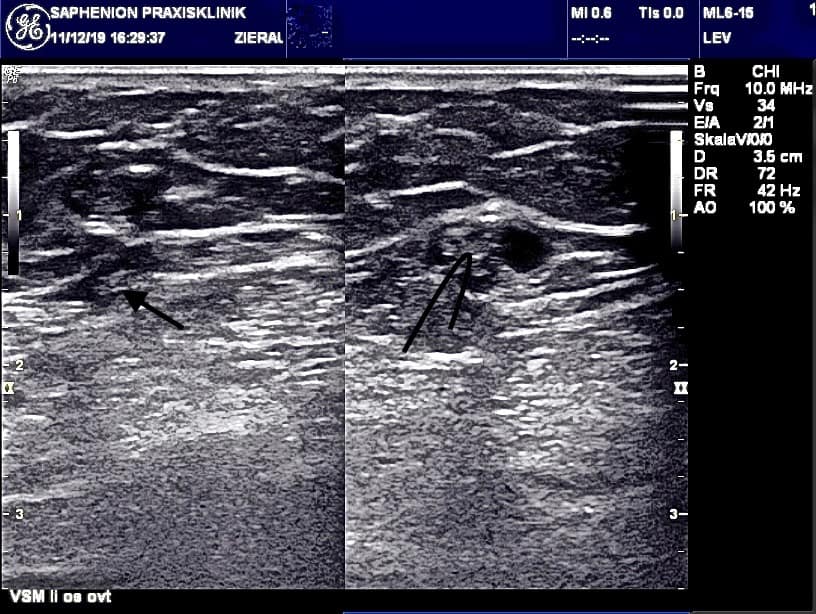
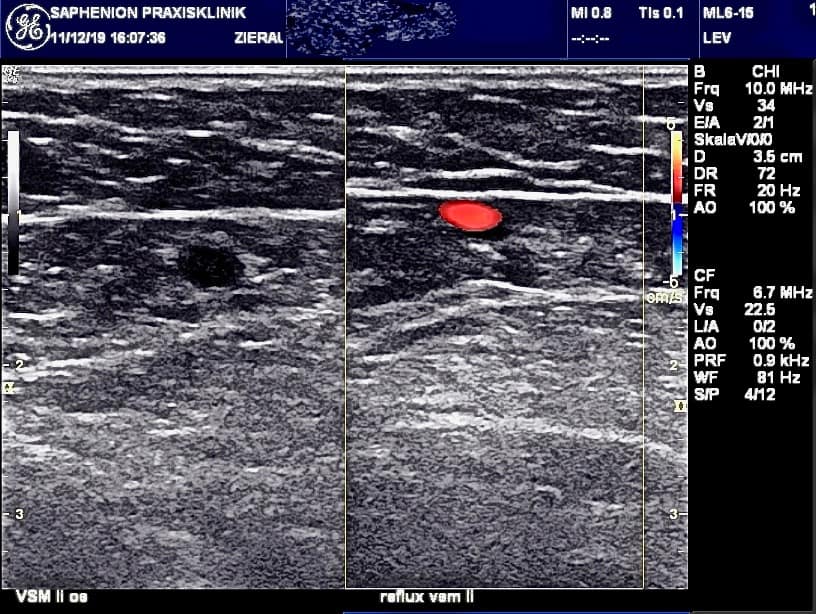
Duplex control follow up over 90 months: the first day 2595 veins were re-examined. 2583 veins were sealed initially = 99,54% closure rate.
After 14 – 30 days we found 50 partial and 15 completely recanalized veins able to follow up of 2585 sealed truncal veins (99,60%) = 97,49% closure rate.
Three to four months post operationem we had 54 partial and 23 complete recanalisations able to follow up 2004 veins (77,2%) = 97,03% closure rate.
After 8 to 10 months we observed 65 partial and 39 complete recanalisations able to follow up 1694 veins (65,3%) = 95,99% closure rate.
No further recanalisations were found after 90 months up to now! The closure rate over all the time of 90 months = 95,99%.

SaphenionScience – 90 months vein glue: technique and side effects.
For the treatment of one truncal vein we need 1.4 ml to 1.8 ml of glue. The time of treatment for one GSV by experienced specialists is 10 – 15 min., for two veins 25 – 30 min.
Unspecific inflammatory reaction of tissue in 191 veins (7,4% truncal veins – no phlebitis!). Bleeding for> 12h ex punctio in 23 cases (continued anticoagulatio perioperative). Lymph-fistula at the puncture site in 19 cases (0,73%).
Glue(?) pimpel = (Granuloma) with cutaneous perforation 10 – 12 months after sealing in 3 cases. Open this pimple with incision, made a histopathological examination – without any glue inside! The pimpels all were healed after 14 days.
There was not to be found: sensory or motor nerv damages, no hyp-/ paresthesia, no allergy, no permanent skin reaction, no phlebitis or thrombosis, no embolism or other heavy side effects.
SaphenionScience – 90 months vein glue: Our experiences and discussion
VenaSeal is one of the endovenous techniques with a high standard of effectiveness – quality standards are the same like other procedures. VenaSeal treatment of all truncal veins, side branches and perforator veins until a diameter of 1,5 – 2 cm is real. Higher diameters are possible! We now don`t see advantages in thermal therapy options.
We also recommend vein glue for ectatic veins (over 1.5 cm in diameter), aneurysms, and postphlebitic truncal varicose veins. Separate punctures with special wing cannulas are also possible here to effectively close the aneurysm sac.
VenaSeal is first choice in treatment of truncal veins of the lower leg and truncal veins of GSV 3 – 4°. VenaSeal is the expensive of all endovenous techniques, thats why therapy of more than one truncal vein simultaneously is recommendable. We can treat in normal 2 – 3 truncal veins with one catheter – Six truncal varicose veins were also closed simultaneously in one session, depending on the severity and anatomy. However, the simultaneous therapy of several truncal varicose veins also reduces the costs compared to classic radical surgery. Here it is quite clear that the thermal systems (Laser, Radiowave, Microwave, are more likely to be used simultaneously with a maximum of 1 – 2 truncal veins.
The actual in the health care market often discussed thermal SONOVEIN – System is actual to be used for one truncal vein. SONOVEIN will once again mean a significant increase in costs for patients and doctors as equipment and for the transducers.
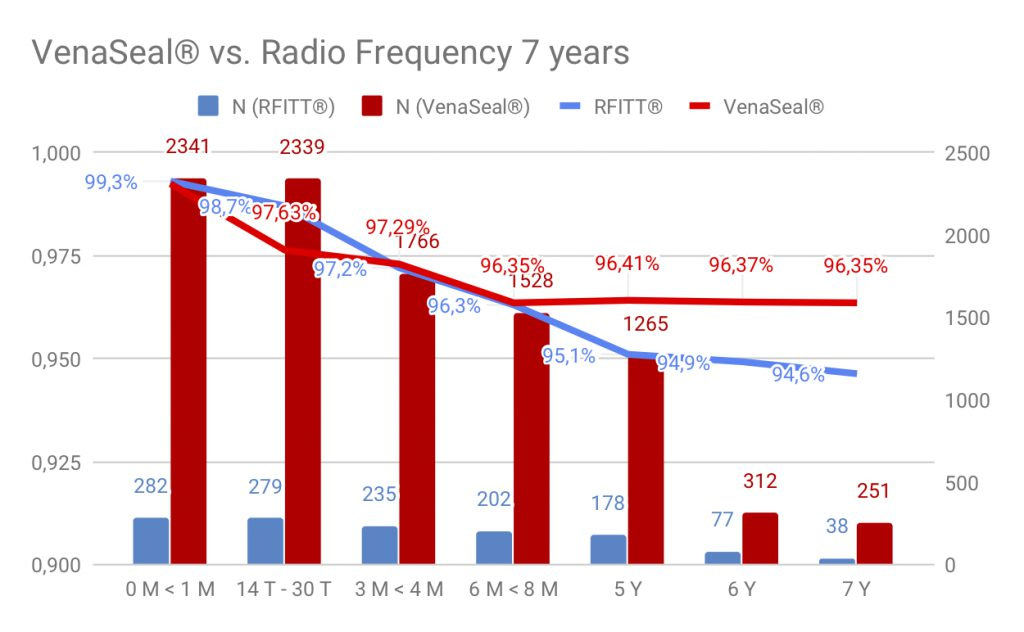
Closure rate of VenaSeal® compared to radiowave / laser is same or better. In comparison of the closure rates of VenaSeal® to radio frequency therapy over a long period of 7 years, the vein glue shows a higher effectiveness in the closure rates.
VenaSeal® works without mechanical or thermal energy, side effects of laser or radio wave are not an issue here. We have no long-term radio wave or laser long-term studies that extend beyond 5 years. The sealing of varicose veins over 1.5 cm in diameter is also effective.
The painscore of sealing veins is significantly lower (1.3 VenaSeal – 3.4 radio frequency) than in radiofrequency or laser therapy. It remains to be seen to what extent the new thermal procedure SONOVEIN Echotherapy shows the same results.
Neither general anesthesia nor tumescence is required, as a rule postoperative compression therapy is not necessary either. This is – in the moment – not possible with the thermal system Laser, Radiowave or SONOVEIN Echotherapy (self test).
SaphenionSience – 90 months vein glue: Our resumes
After 18 years of working with endovenous techniques, the conclusion: Our experience with the endovenous therapy and especially with the cyanoacrylate vein glue VenaSeal® make it our basic therapy for truncal varicose veins. Radical surgical therapy is only necessary for rare exceptional situations. In this respect, different statements of experts, colleagues, and health insurance companies are urgently critical to question after a long time of use (8 years). Thermal catheter therapy is not out, but the endovenous pathway has become more sophisticated! Noninvasive SONOVEIN Echotherapy is also a thermal procedure with the associated side effects but does not require catheters or vascular puncture. However, thrombosis prophylaxis and tumescent anesthesia is also indicated along the vein to be treated during the SONOVEIN therapy.
In addition to the venaseal treatment of varicose veins, we will continue to use a – currently considerably modernized RFITT radio wave system (veins, baker cysts).
The targeted introduction of the SONOVEIN system is currently deferred for clinical and commercial reasons. The indications for SONOVEIN and its advantages over the VenaSeal or the available minimally invasive thermal catheter procedures must first be precisely defined by the physicians.

Photos / Videos
Claudia Reuter, Utzius
Paintings:
Gustavo – Collection Saphenion
Links / Papers:
Gibson et al.: Twenty-four-month results from a randomized trial of cyanoacrylate closure versus radiofrequency ablation for the treatment of incompetent great saphenous veins.
Ivanova, Patricija: Post procedere neuropathy: comparison of surgery, EVLA and glue; Presentation on 2nd. NEEF, Riga, 17th.August 2019.
Lawson J, S Gauw, C van Vlijmen, P Pronk, M Gaastra, M Mooij, C Wittens: Sapheon: the solution? Phlebology 2013;0:1-8
Morrison, Nick, and Kathleen Gibson: Veclose Study: Preliminary Month 1 Data; 2nd Annual Cyanoacrylate Embolization Symposium, Mainz, Jan. 18, 2014
Proebstle TM, Alm J, Rasmussen L, Dimitri S, LawsonJA, WhiteleyM,, Franklin IJ, Davies AH: The European Multicenter Study On Cyanoacrylate Embolization Of Refluxing Great Saphenous Veins Without Tumescent Anaesthesia And Without Compression Therapy. eScope – study; Abstract presented to the American Venous Forum Annual Meeting 2013, Phoenix ( AZ) the USA
Rasmussen LH, Bjoern L, Lawaetz M, Lawaetz B, Blemings A, Eklof B.: Randomised clinical trial comparing endovenous laser ablation withthe stripping of the great Saphenous vein: clinical outcome and recurrence after 2years. Eur J Vasc Endovasc Surg 2010;39:630 – 35
Thum, J: Single Center Erfahrungen aus 234 Stammvenenbehandlungen mit viskösem n – Butyl – 2 – Cyanoacrylat- Verwendbarkeit, Akzeptanz, Ergebnisse; vasomed 1, 2019; 28 – 31
Üdris, Ints: 6 years single center results „Baltic Vein Clinic“ of truncal varicose vein sealing; Presentation on 2nd. NEEF, Riga, 17th. August 2019.
Zierau U Th.: Venenverklebung versus Radiofrequenztherapie bei Varicosis – Verlaufsstudie über 36 Monate mit 1139 Behandlungen. vasomed 28 (2016) 212 – 216.
Zierau U Th.: https://www.saphenion.de/news/konsensus-1-neef-endovenoese-krampfadertherapie.
Jones AD, Boyle EM, Woltjer R, Jundt JP, Williams AN. Persistent type IV hypersensitivity after cyanoacrylate closure of the great saphenous vein. J Vasc Surg Cases Innov Tech. 2019 Aug 7;5(3):372-374. doi: 10.1016/j.jvscit.2019.05.004. eCollection 2019 Sep. PubMed PMID: 31440717; PubMed Central PMCID: PMC6699189
Navarro-Triviño FJ, Cuenca-Manteca J, Ruiz-Villaverde R. Allergic contact dermatitis with systemic symptoms caused by VenaSeal. Contact Dermatitis. 2019 Oct 31. doi: 10.1111/cod.13431. [Epub ahead of print] PubMed PMID: 31674037.
Almeida JI, Murray SP, Romero ME. Saphenous vein histopathology 5.5 years after cyanoacrylate closure. J Vasc Surg Venous Lymphat Disord. 2019 Jul 4. pii: S2213-333X(19)30325-7. doi: 10.1016/j.jvsv.2019.04.014. [Epub ahead of print] PubMed PMID: 31281102.
Park I, Kim D. Correlation Between the Immediate Remnant Stump Length and Vein Diameter After Cyanoacrylate Closure Using the VenaSeal System During Treatment of an Incompetent Great Saphenous Vein. Vasc Endovascular Surg. 2019 Oct 3:1538574419879563. doi:10.1177/1538574419879563. [Epub ahead of print] PubMed PMID: 31581906.
http://evtoday.com/2017/04/three-year-veclose-data-and-1-year-waves-data-presented-for-medtronics-venaseal-closure-system
https://www.saphenion.de/news/sealing-lower-leg-truncal-veins-kleben-von-stammkrampfadern-am-unterschenkel/J Vasc Surg Venous Lymphat Disord. 2018 Sep;6(5):606-613.
http://www.ncbi.nlm.nih.gov/pubmed/?term=veclose
http://www.ncbi.nlm.nih.gov/pubmed/27052039
http://www.ncbi.nlm.nih.gov/pubmed/23482526
https://vsearch.nlm.nih.gov/vivisimo/cgi-bin/query-meta?query=venaseal&v%3Aproject=nlm-main-website
http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfmaude/detail.cfm?mdrfoi__id=4841327